Eastern Great Egret
Ardea modesta (Gray)
Ardea modesta J. E. Gray, 1831. Zool. Misc., p. 19: India.
Other names: Great Egret, Large Egret, White Egret, White Heron, Kotuku (of Maori origin in New Zealand), White Crane, Large Heron in English; Garceta Grande in Spanish; Grande aigrette in French; Большая белая цапля in Russian; Kuntul putih besar in Indonesian; Tagak, Talabong in Pilipino; Dai-sagi in Japanese; Da bailu in Chinese.
Description
The Eastern Great Egret is a large white heron with yellow bill, dark legs, and in breeding, long back plumes.
Adult: The base color is entirely white. The head lacks plumes. The bill is yellow with grey black tip. Lores are yellow. Iris is yellow. A black line of bare skin the gape extends beyond the eye. The back lacks breeding plumes. Legs and feet are brown to grey black, two-toned, the upper leg being lighter than lower leg.
During breeding, the long straight (not recurved) aigrette plumes extend beyond the tail. The crest and neck have slightly long, loose feathers but not plumes. During courtship the bill turns grey black. In courtship there appear to be short term flushes of color, especially the irises that can turn red. The lores are variably pale green to olive green. The whole legs and sometimes the feet can take on a reddish wash over the otherwise dark legs and feet, the color being variably pink, red, purple red or brown red. After courtship, during incubation and chick rearing, the bill returns to yellow starting from the base leading to bicolor condition; the irises return to yellow; the lores fade to dull green; legs and feet change to black.
Variation: Sexes have similar plumage. Males are larger than females. Birds from New Zealand (sometimes called maoriana) are reported to be larger than birds from China and India.
Juvenile: Juveniles are similar to adults, but lack ornamental plumes. They have yellow bills with a blackish tip.
Chick: Nestlings have cream white down. Their bill is black tipped with yellow, and a gape that extends beyond the eye. The iris is straw colored. The lores are green. Longer down on the head forms a crown. The legs are grey.
Voice: “Cuk” call is the disturbance call given at the nest, rendered “cuk, cuk, cuk, cuk”, or “glok, glok, glok, glok”. The “Arrrr” call is the disturbance call uttered when taking flight, rendered “ar, ar, ar, ar, aaar”. It is probably equivalent to the “Kraak” call attributed to the Great Egret. The “Gorork” call, the Greeting Ceremony call, is a “gorork, gorork, gorork” followed by rapid, repeated croaking “Grock”. This call seems very different from the “Rrrooo” call of the Great Egret. Young beg with a “Kek” call, rendered “kek, kek, kek, kek.”
Weights and measurements: Length: 83-103 cm. Weight: males 970 g, females 760 g.
Field characters
The Eastern Great White Egret can be easily confused with the other white herons. It is identified by its size, bill size and color, back plumes (in breeding), long kinked neck, and extension of a dark line of the gape from the corner of the bill to beyond the eye.
It is distinguished from the Great Egret of the west by its slightly smaller size, two tone brown to grey black legs and yellow bill and in breeding by a combination of black bill, green lores, pink to purple red wash on dark legs. It is distinguished from the Intermediate Egret by its somewhat larger size, relatively longer (not stubby) bill, relatively longer, thinner more kinked neck, relatively longer legs, lack of obvious head crest, the line of its gape (extending beyond not ending at the eye), and in breeding its back plumes and red-washed legs. It is distinguished from Little Egrets and Chinese Egrets by its larger size, yellow (not black) bill except in courtship, dark (not yellow) feet, and long neck. It is distinguished from Cattle Egret by its larger size, height, long neck, and lack of buff color. It is distinguished from white Eastern Reef-Herons by its larger size, tall, slim appearance, long neck, and in breeding its back plumes.
Systematics
In this chapter we recognize the eastern form of the great egret, long classified as Egretta alba modesta, as a separate species in the genus Ardea. Treating first the issue of genus, one of the more critical recent findings about heron systematics using molecular techniques is the confirmation of morphological evidence that Ardea and Egretta are not necessarily sister taxa and that there are, in fact, character gaps between the two groups of birds (Payne and Risley 1976, Sheldon 1987, Sheldon and Slikas 1997, Sheldon and Whittingham 1997). In this light, the congruence of morphological, behavioral, and biochemical evidence leaves little doubt that the birds known as great egrets birds are not Egretta but are more closely allied to Ardea.
At the specific level using molecular evidence, Sheldon (1987) found clearly that the south east Asia population of great egrets is distinct from the North American population. The degree of distinction is equivalent to that of the Intermediate Egret and so should be recognized equivalently, i.e., as separate species. Beyond molecular evidence, the Eastern Great Egret is differs from the western Great Egret in having red legs during nesting (Hancock 1984b) and using an Aerial Stretch display. In courtship it uses the Stretch as an advertising display but not also as a greeting display and its Snap lacks a heavily aggressive component, behavioral trait showing similarity to Egretta (Hancock 1984a, Voisin 1991).
Range and status
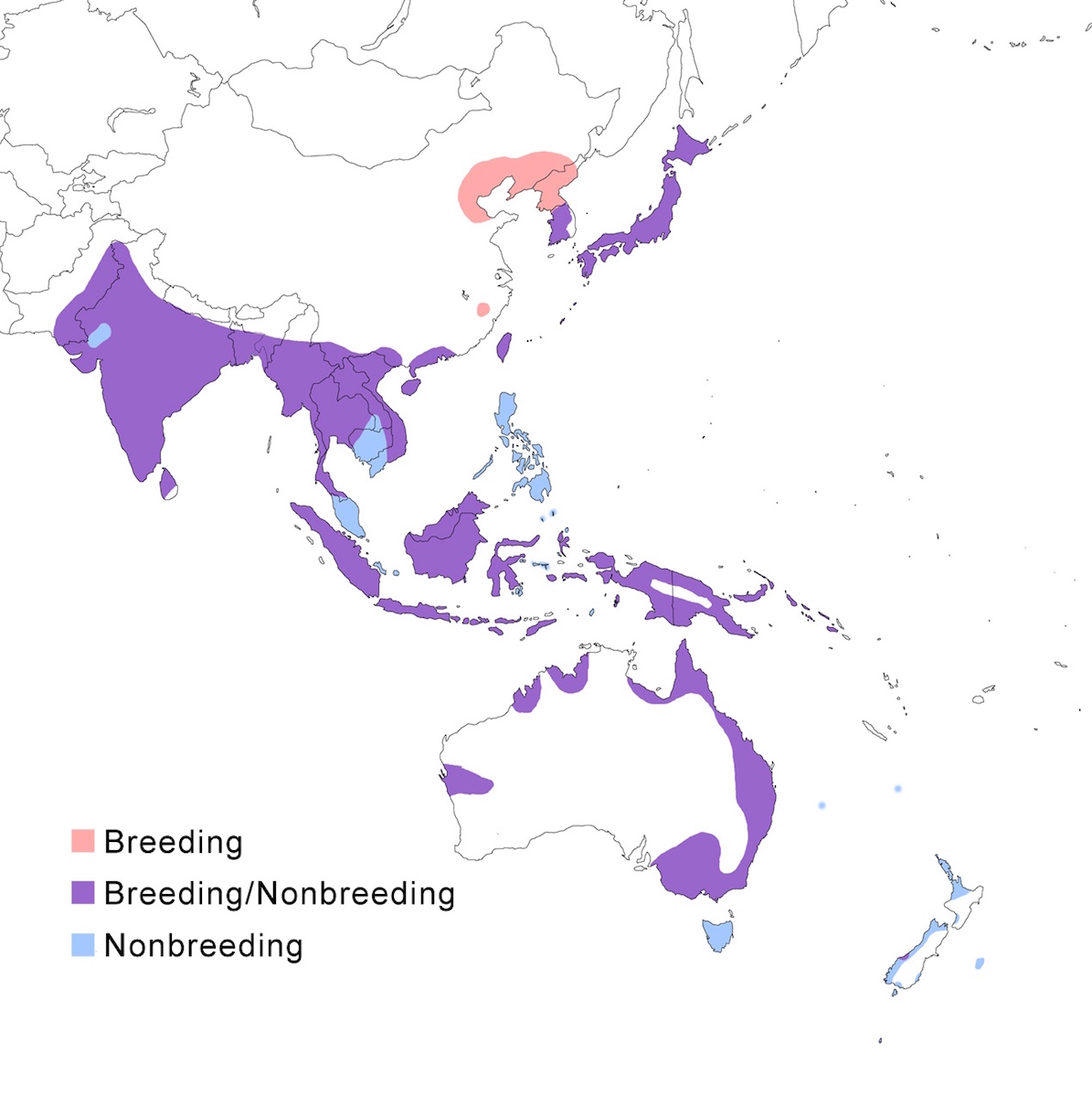
RANGE: The Eastern Great Egret occurs in south and east Asia and Australasia.
Breeding range: It nests in Pakistan, India, Sri Lanka, Bangladesh, Myanmar, Thailand, China (south east Heilungkiang, Shantung Peninsula, Fukien, south Yunnan), Korea, north east Russia (south Ussuriland), Japan (Kanai 1990, Narusue 1992, Ueda 1994), Indochina, Indonesia (Sumatra, Java, Borneo, Sulawesi, Lesser Sundas, Moluccas, Iran Jaya), Papua-New Guinea, Solomon Islands, Australia (except drier interior) and New Zealand (a single colony on South Island). There is not now breeding known in Malaysia or Philippines (Lansdown et al. 2000).
Nonbreeding range: It is a summer visitor to Japan as far as Hokkaido and the Kurils (Kosugi 1993, Iijima 1984, Shimada et al. 1994). It occurs in nonbreeding season from extreme south Japan (Honsu, Ryukus Islands), south east China, Hainan, and Taiwan, through the East Indies, Australia and New Zealand
Migration: Post breeding dispersal is characteristic of Eastern Great Egrets. Within Australia birds disperse in all directions but tagged birds have been recorded returning to the natal colony to breed (Geering et al. 1998). Dispersing birds appear on distant islands, such as Macquarie, Lord Howell, Chatham and Norfolk islands, Fiji (Suva) (Mackereth 1992). Red-legged Great Egrets seen in Yugoslavia and in the Netherlands have been attributed to vagrant individuals of modesta (Ouweneel 1982, Gaasbeek 1982, 1985, Hancock 1984a). Although birds in Indochina through Indonesia are apparently resident, portions of Asian and Australian populations migrate. The northeast population migrates from Korea to Japan to the Philippines. In Australia, inland nesting birds migrate through Queensland (Geering et al. 1998) to New Guinea and southern birds to New Zealand.
Status: The species is widespread and common to abundant over much of its range including India, Australia, and New Guinea (Lansdown et al. 2000, Maddock 2000). Counts include almost 11,000 in India and 5,000 in the Philippines. The single nesting site in New Zealand supports a stable nesting population of about 100 birds (Miller 2001), owing in part to the increase in fish ponds and protection of the site (Maddock 2000). Recent surveys of two areas suggest it is very scarce in China (M. Fasola pers. comm.).
Habitat
The Eastern Great Egret uses a variety of habitats, preferring permanent water of pools and streams in flood plains, lake and river margins, swamps and marshes (Eleocharis, Typha, Phragmites, Scirpus), and seasonal herbaceous marshes. Along the coast, it uses mud flats, lagoon, mangrove swamps, tidal streams, salt marsh, salt pans, and off shore reefs. Colony size has been related to the available area of salt marshes (Baxter and Fairweather 1998). It also uses human altered habitats including reservoirs, sewage ponds, flooded fields, pasture, and ditches (Young and Chan 1997).
For nesting it requires isolated trees or bushes, usually surrounded by or near water. Its favoured areas are wetlands with tall trees. It uses mangroves along the coast, inland swamps, river edge forests, and lake edges and islands. It also seems to require trees and islands for roosting.
Foraging
The Eastern Great Egret is a diurnal forager, its foraging timing depending on daylight, tides, and weather (Min and Shiraishi 1984a). It feeds singly, in small groups and occasionally in very large aggregations; it feeds by Walking slowly, stalking prey (Recher et al. 1983, Tojo 1996). It primarily Walks slowly in open shallow water about 30 cm deep, using an Erect posture. It also Stands motionless with head extended. It often uses Head Tilting, Peering Over, and Head Swaying. It feeds from the air by Plunging. Swimming has been reported (Viney 1988). When feeding as part of aggregations, it frequently engages in antagonistic behavior to defend its feeding spot or take prey from other birds such as cormorants. It is also robbed of prey and feeds commensally by following other birds in the aggregation (Maddock 1992).
Sexually oriented aggression has been reported on the feeding ground. One bird in Hong Kong in spring migration advanced on other feeding birds in stiff legged fashion neck outstretched, bill pointing to 45 degrees, and back feathers erected. The importance of feeding ground display is undetermined.
Prey taken is extremely diverse but principally fish. It also takes frogs, invertebrates, and even small birds. Invertebrates include crickets, dragonfly nymphs, flies, beetles, snails, bugs, crayfish, and prawns. Among the birds recorded are chickens, crake (Porzana), kingfisher (Halcyon), sparrow (Passer) and white eye (Zosterops), swallows (Hirundo), tern (Sterna) (Peter 1994, Darnell and Darnell 1995). Prey taken is relatively small, under 12 cm, probably as a way to avoid being pirated by eagles (Haliaeetus), kites (Haliastur), or other wading birds.
Breeding
The breeding biology of the Eastern Great Egret has been well studied (Min and Shiraishi 1984a, b, Min et al. 1984a, b, Maddock 1986a, b, Maddock and Baxter 1991). Nesting timing in Australia is variable depending in part on rainfall (Geering 1993), but generally from November to early May. In Korea, nesting is from April to August (Min et al. 1984b). This egret nests in reed beds, trees or bushes, usually in isolated places or on islands. Trees used include larch (Larix), mangroves, paper bark (Melaleuca), eucalypt, and iron wood (Casuarina).
The Eastern Great Egret is primarily a colonial nester, usually with other species including other herons such as night herons (Nycticorax), ibises, spoonbills, and cormorants. It places its nests relatively high in the vegetation. The nest, made of sticks, is shallow platform and usually less than 1m in diameter. The nest is built almost entirely by the female, using sticks brought by the male.
The bird is highly colonial. Large numbers accumulate at the nesting site, and the male claims and defends its display territory. The Stretch is its principal advertising display. In the Stretch display, the head is thrust vigorously upwards, followed by withdrawal and equally vigorous strong downward bob of the legs (M. Maddock pers. comm.). Wing Preen, Twig Shake, Circle Flight and Bill Clappering with bills in contact have been observed during and following pair formation. Long sequences of repeated Wing Preens and violent Twig Shakes are interspersed with Stretch displays is a typical behavioral series (M. Maddock pers. comm.). An Aerial Stretch display has been documented in modesta, but has not been recorded in Ardea alba (Hancock 1984b). Mock Preening and Allopreening occur after pair-formation. In the Greeting Ceremony, the bird erects its display plumes and raises its wings giving the Gorork call.
The eggs are pale blue to pale blue green, 52 x 36 mm. The clutch size is 2-5, averaging 3.8 in Korea. Incubation is accomplished by both parents but mainly by the female who incubates for 80% of the time. Incubation begins after the first egg and lasts 23-29 days.
Young are semialtricial and are tended by both parents. Feeding is first at the nest and later away from the nest. Older chicks grab the adult’s bill to force regurgitation. In the early stages of chick rearing, one parent remains at the nest on guard. The elaborate Greeting Ceremony continues to be performed at the changeover. In the latter stages of nesting, the young are left unguarded (M. Maddock pers. comm.). Little information is available on the details of chick development. Nesting success is often related to hydrological conditions (Maddock 1986a, b, Maddock and Baxter 1991). High chick mortality is also due to starvation following death of parents and loss of chicks due to sibilicide during dry seasons has been observed (M. Maddock pers. comm.). Nests built in inappropriate trees are subject to loss (Min and Shiraishi 1984b) and only 40% on these hatched eggs. At a colony in Australia, 91% of nests fledged some young averaging 2.1 chicks per successful nest.
Population dynamics
Tagged birds have been recorded returning to their nesting colony as two-year old birds (M. Maddock and D. Geering pers. com). Adult mortality during the breeding season was 5.8-7.2% during April–June, probably reflecting energy deficits due to spring migration, stresses of breeding and pesticide poisoning (Min et al. 1984b).
Conservation
The species, in Australia and New Zealand, has benefited from irrigation schemes creating foraging sites. Its tendency to feed in rice fields makes it dependent on this habitat is some areas, such as Japan. Changes in rice culture may adversely affect use it, and this important issue requires additional study (Narusue and Uchida 1993, Subramanya 1996, Lane and Fujioka 1998). Protection and management of feeding sites are essential. Its natural habitat in Australia remains under constant threat from drainage, grazing, clearing, burning, salinization, and exotic plants. As a result, there has been a serious decline (80%) in the breeding population at eastern NSW nesting colonies 1988-89 to 1997-98 (Maddock 2000), with no evidence of a recovery since (M. Maddock pers. comm.). Decline has also occurred in Victoria, where its Conservation Classification is endangered (NRE 2000).
It is flexible in its nesting sites and can even nest near residential and industrial settings. But nesting colonies, whether on natural or artificial sites, need to be protected and in some cases actively managed (Min and Shiraishi 1984b, Baxter and Fairweather 1998). For example, appropriate trees need to be planted for both short-term and long-term use. In Australia, current moves to provide environmental water flows from regulated rivers to breeding colonies in the Murray-Darling inland river system (Maddock 2000) have the potential to halt population declines there. However, the threats to the species are not only confined to the breeding colonies but also to habitat along the migration routes, which must also be addressed.
Research needs
Needless to say, interesting taxonomic issues remain within great egret populations. Our recognition of eastern populations of great egrets as a separate species deserves critical examination. A study of geographic variation in size and soft-part colours of all eastern great egret populations is needed to decipher the relationships among Australian egrets, and the egrets of mainland Asia. Attention needs to be focused on the breeding situation in the area of potential overlap in north China and in Japan, where birds considered to be intermediate between albus and modesta have been described (Amadon and Woolfenden 1952). Analysis is confounded by the presence of breeding birds, winter migrants, and summer dispersants in these areas. The question also remains as to whether the European and African forms are more closely related to the North American egretta or Australasian modesta, or even constitute separate species themselves.
The specific courtship of the Eastern Great Egret has not been studied in sufficient detail. Given the importance being placed in its own species, partially due to the existence of an Aerial Stretch display and breeding leg coloration, there is a need to better document courtship behaviour, nesting behaviour, colour changes, and vocalizations of modesta. Banding studies are needed to further decipher the range of the species, its migration patterns, and important non-breeding areas.
Overview
The Eastern Great Egret is a bird of shallow wetlands, where its slow, patient stalking and waiting behaviour can be effective. It catches relatively small fish and other prey, and so feeds for long periods during the day. It is primarily solitary in its feeding, which avoids disturbing its prey and competing with others. When feeding at concentrated food sources, it is highly aggressive, so much so that feeding appears to be interrupted. It can use a variety of feeding locations, from reefs to pasture including artificial situations, from reefs to pasture including artificial situations. Despite its solitary preferences in feeding, it is highly colonial in nesting. Much remains to be observed about the basic biology of these species, especially in comparison with other large white egrets.