Goliath Heron
Ardea goliath (Cretzchmar)
Ardea goliath Cretzchmar, 1826. Ruppells Atlas Reise (Vogel), p. 39, p1.26: ‘Bahhar Abiad’ = Bahr el Abiad or White Nile.
Other names: Garza Goliat in Spanish; Héron goliath in French; Goliathreiher in German; Goliathreiger in Dutch, Goliathägar in Swedish; Цапля-голиаф in Russian.
Description
The Goliath Heron is a very large grey heron with chestnut head, neck and belly. It is the largest living heron.
Adult: Its head is red chestnut, with an elongated, bushy chestnut crest. The bill is very large and thick, black above and pale horn below. The irises are yellow rimmed in red, and the lores are green yellow. Sides of the face and neck and back of the neck are a pale chestnut. The chin and throat are white, with black streaks extending down the neck onto the upper breast. Its back and upper wings are slate grey, with a chestnut “shoulder” patch at the bend of the wing when closed. Lanceolate plumes on the and back are slate grey. The upper breast has white lanceolate plumes that hang over the deep rufous chestnut of the breast and rest of the under parts. Under wings are pale chestnut. The relatively long legs are black, as are the feet.
Variation: Females smaller, with slightly smaller bill. There is no recognized geographical variation, although if there remains an isolated population in Bangladesh geographic variation is probable.
Juvenile: Juveniles are the same as adults, but the chestnut is paler and more rufous. Grey areas are tinged with brown. Undersides are mottled with black and white striping on neck less defined. It lacks elongated plumes.
Chick: Chicks are covered with white grey down. Iris is light green, bill brown, legs and skin lime green (Cooper and Marshall 1970).
Voice: Goliath Herons have a distinctive “Kowoork” call, a loud, repeated, individually distinctive bark audible for up to 2 km. A sharp “Arrk” call is the disturbance call. ”Krooo” call is given during Stretch at the vertical position whereas a “Huh-huh” call is given at the crouched stage (Salewski and Grafe 1999). “Organ like dueting” has been reported at nest (Brown et al. 1982), but more details are needed to confirm this.
Weights and measurements: Length: 150 cm. Weight: 4,300-4,468 g.
Field characters
The Goliath Heron is identified by its great size combined with its grey and chestnut coloration. It flies with slow, deliberate wing beats, with the wings markedly bowed. When the bird takes flight its head and neck are at first outstretched, but soon retracted onto the shoulders.
It is distinguished from the Purple Heron by its large size (nearly twice as large), chestnut rather than black cap, and chestnut rather than black-streaked rufous sides of neck. It is distinguished from the White Bellied and Sumatran Herons by its grey and chestnut plumage (not solid grey or grey brown) and the lack the black and white neck pattern.
Systematics
This is one of the great herons, and is probably most closely related to the White Bellied and Sumatran herons.
Range and status
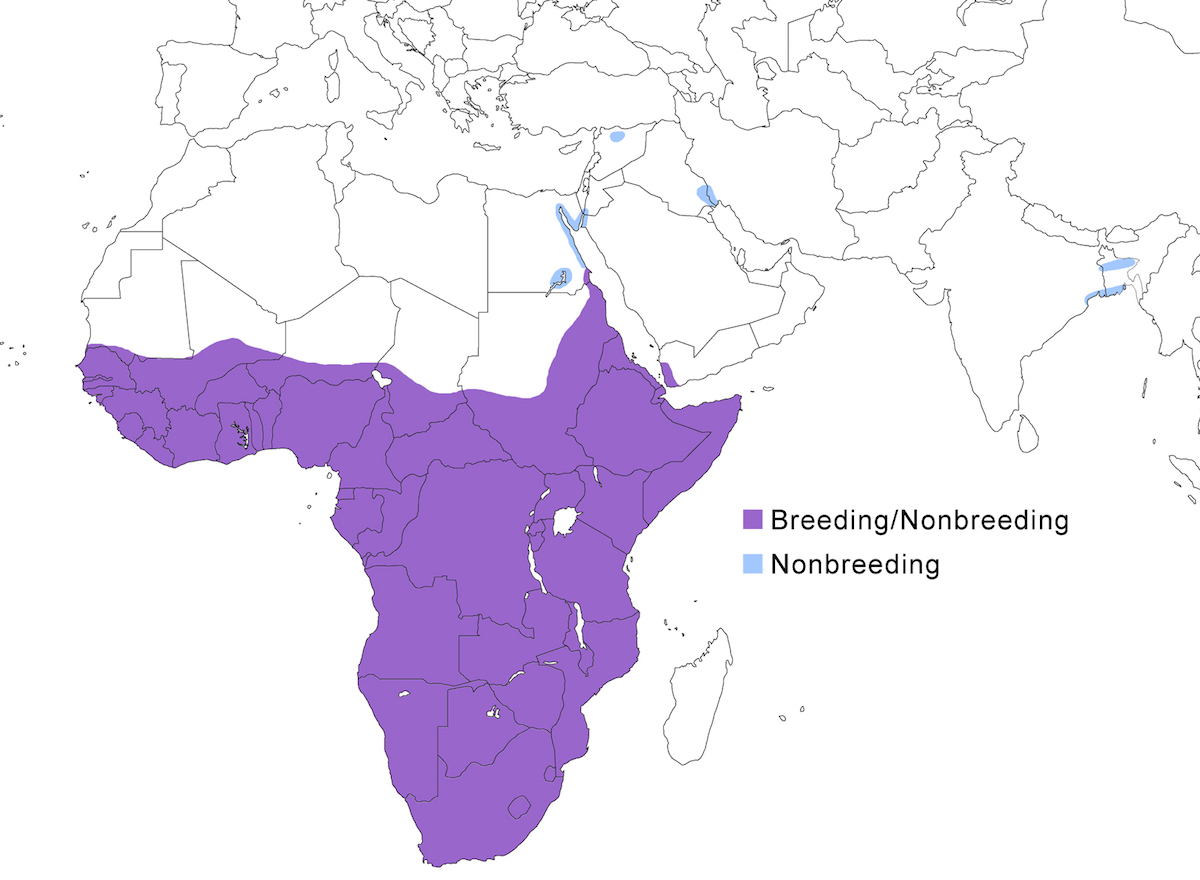
The Goliath Heron is a bird of Africa and the Middle East, and also the Indian subcontinent.
Breeding range: The Goliath Heron breeds in suitable habitat over much of Africa. It occurs along the east coast from Egypt and Sudan (Goodman and Storer 1987), Yemen on the Red Sea (Porter and Al-Saghier 1998), Somalia (Schels 1993), to Cape Province South Africa (Pauw 1991), and inland Africa west to the south west deserts, Gambia, Guinea-Bissau, north to the Sahara. Their specific breeding locations within this larger range are not so well documented.
Nonbreeding range: Birds occur commonly along both sides of the Red Sea (Egypt including Sinai, Sudan, Saudi Arabia, Yemen) and has been increasing there over the past decades (Bjlsma and de Roder 1982). In this area, breeding is known from northern Sudan, south east Egypt, and probably Yemen (Goodman and Storer 1987, Brooks et al. 1987, Porter and al Seighier 1998). Birds also occur, or have occurred in the past, in Syria, northeast Israel, south Jordan, south Iraq, and Iran (Kinzelback 1986, Kasparek et al. 1994). The species has nested in the past in Iraq, but there are not recent records it also has been seen several times recently in Bangladesh (in the Sundarbans) and India (Assam) (Saikia and Bhattacharjee 1990, Thompson et al. 1993, Madge 1995). It formerly occurred elsewhere in India and Sri Lanka. Given the distances involved to the core of the known range, it may be that these birds represent a small population breeding in the Sundarbans or Assam, but this needs to be documented. There is also an old record from Madagascar in 1879, but none since and a misidentification should not be ruled out.
Migration: The species is sedentary and regular migration is not documented. However local movements occur in response to foraging conditions. Origin of birds outside the known breeding range needs to be assessed to determine if dispersal occurs.
Status: No figures are available on population sizes. The species is locally common in east Africa swamps and along some lakes and rivers (Turner 2000). But over most of Africa, it is widespread, thinly spaced. An estimate for Tanzania is 2,500-4,000 birds (Baker and Baker in prep.). For example they average only about one per 4 km2 along a river course (Dowsett and de Vos 1964). It has increased in South Africa due to dams and reservoirs (Geldenhuys 1984), has expanded its range in Cape Province, South Africa (Pauw 1991), and is increasingly seen along the Red Sea. However, population status of birds in Iraq-Iran and the Indian subcontinent remains unclear but is presumed to have severely declined in both areas.
Habitat
The Goliath Heron is a very aquatic heron of both coastal and inland habitats, rarely wandering far from water. The shallow water margins of large lakes, lagoons, and large river systems are its favored habitat. It typically feeds out from shore in deep water and near floating plants. Over all though, it inhabits many types of shallow waters including rivers and lakes, both salt and fresh, marshes and swamps, tidal estuaries, reefs and sometimes mangrove creeks and even water holes in woodland savanna, from sea level to 2,100 m. It is not commonly found around areas of human disturbance.
Foraging
The Goliath Heron is typically a solitary hunter defending large feeding territories from other Goliath Herons (Whitfield and Blaber 1978, Mock and Mock 1980). Occasionally two may be seen together. It flocks very rarely. It typically feeds diurnally, but also feeds at night (Hustler and Games 1987). The usual hunting method, day or night, is Standing in shallow water or on floating vegetation, grassy shores or coral. It Stands for long period, seemingly immobile waiting for a prey item to appear. Because of its size, this heron can wade well away from shore. Typically, it begins to hunt by adopting a very Upright posture, but the head is soon lowered and held horizontally and the neck crooked ready for its thrust. Head Tilting is used infrequently, and it is possible that the bill may be used to attract fish. After long intervals of patient waiting, it may move to a new area by Walking Quickly or Hopping. When it flies to a new site, it often lands on the floating vegetation rather than the open water. It also Walks Slowly, in a very stealthy manner. It fishes along the coast on rising tides.
Fish are caught by a lunging Bill Thrust, catching fish deep in the water, normally spearing them with mandibles held slight apart so as to have both run through the prey. They are brought to the surface and shaken off the bill before being killed by restabbing, beating and poking with the bill. Herons often place their prey on the tops of floating plants while the subdue it. Handling time is high and one-quarter may then be lost, either escaping on their own or through piracy by other fish-predators. Fish Eagles (Haliaeetus) are frequent harriers of Goliath Herons (e.g., Abbott 1994), but also pelicans (Pelecanus) and Saddle-billed Stork (Ephippiorynchus). Herons defend their territories using Forward, Fluffed Neck, Upright, chasing and supplanting.
The food consists almost entirely of fish. It is a specialist in large fish, taking a few fish each day (2.3 in one study) and ignoring smaller ones. In Natal the average length of fish caught was 30 cm weighing 500-600 g, but one of 50 cm was too big to swallow it. River bream (Acanthopagnus), mullet (Mugil) and tilapia (Sarotherodon), and barbel (Barbus) are among the fish taken. They also will eat other things that come their way, including prawns, frogs, lizards, snakes and small mammals.
Breeding
The breeding season generally coincides with the start of the rains. In some places, breeding is year round with no discernible peak season. In other situations, such as south Africa, birds may not breed every year.
The Goliath Heron uses a diversity of nesting habitat. The preferred nesting choice appears to be on islands or on islands of vegetation protected by surrounding waters, as suggested by the observation that birds abandon nest sites when an island becomes attached to the mainland. Colonies are usually on islands in lakes and other water bodies. Solitary birds nest on riverbanks, lakeshores, and small islands. It is a big bird and nests low in the vegetation, generally below 3 m. Nest sites include sedge, reeds, Typha, Agave, small trees, low bushes, mangroves, and cliffs. On islands any tree shrub stone or bare ground available is used (Geldenhuys 1984).
Nesting dispersion is variable. They nest solitarily, near colonies, within loose single species colonies, and within mixed-species colonies along with other herons, cormorants, darters, ibis, and gulls (e.g., Dean 1988, Kopij 1996a). Several nests may be placed in one tree, and nests in adjacent tress may be less than 20-30 m apart (Brown et al. 1982). In some areas almost 80% of birds nest in colonies (Geldenhuys 1984), and there is evidence that nesting success may be greater for birds nesting in colonies than nesting solitarily (Dean 1988). Although they do nest in solitary pairs through much of their large range, the former view of this species as fundamentally a solitary nester seems not to be correct.
The nest is a large platform made of sticks or reed stems at least 1-1.5 m in diameter. The nest can be flimsy when materials are limited, such as on islands (Dean 1988). One adult builds the nest alone or both birds of the pair build. The courtship displays of this heron are largely unknown. The rarity of observations suggests that courtship may be subdued, perhaps owing to re-paring of previously paired birds. Stretch display has been recorded and possibly a form of Forward (Brown et al. 1982).
Eggs are pale blue, 64.8-80 x 49-58.9mm. They weigh about 108 g (92-117 g), fresh. The usual clutch is 3 or 4, ranging from 2 to 5. Incubation begins with the first egg by both parents, leading to size difference among young (Cooper and Marshall 1970). Incubation takes 24-30 days. A replacement clutch has been recorded. Hatching success has been reported to be low, about 25% (Dean 1988). The young are fed by regurgitation on to the nest floor, only twice daily. After hatching older young can trample younger siblings, leading to brood reduction. Within a few weeks they can scramble out of the nest and defend themselves using Bill Jabbing and size enhancing postures. At about 5 weeks they leave the nest completely. The parents attend them for a variable time, noted to be from about 40 to 80 days. Survival from hatching to leaving the nest was about 62% (Dean 1988). The White-tailed Eagle is a predator in colonies (Ytreberg 1992). Production is 1 or 2 young per successful nest (Brown et al. 1982).
Population dynamics
Nothing is known about the population dynamics and demography of this species. It is likely long lived, with high adult survivorship.
Conservation
In Africa nest sites, particularly colonies, need be protected. Island colony sites should probably be the focus of protection and management (Dean 1988). Feeding sites also need to be protected and managed. More critical to conservation is determining the status of this species as a nesting bird in south Iraq/Iran and the Indian subcontinent. Birds still occur in these areas, and breeding remains likely. Should birds be found, immediate conservation action needs to be undertaken to protect the birds, their nest site, and feeding habitat.
Research needs
Much is known about the feeding biology and general nesting biology of this species. However, detailed information on courtship behaviour, pair formation, the identity and history of paired birds, nesting success, survival, and dispersal is needed. Long-term banding studies are desirable. The status of the species in the Middle East and Indian sub-continent needs to be determined. Surveys are needed in the Middle East, Bangladesh, and can India to determine the distribution, population status, important sites, and nesting situation of this species.
Overview
The Goliath Heron is a unique heron due to its large size and its dependency on very large fish. It has a suite of adaptations to accommodate this role. Its large bill can handle large fish. It legs, relatively long even for its great size, allow it to access deeper water where the large fish are. Its solitary habits likely are required to access this scarce resource. Catching large fish is neither frequent nor efficient, and handling becomes even more problematical. However, its low-key foraging technique conserves energy. It nests on or near the ground or low vegetation, probably by necessity of its size, and this requires protected sites. It is a very aquatic heron, sparsely scattered in the landscape, selecting shallow, protected waters having large fish.