Reddish Egret
Egretta rufescens (Gmelin)
Ardea rufescens Gmelin, 1789. Syst. Nat. 1, pt 2, p. 628, ex Daubenton p1. Enl. p1. 902: Louisiana.
Subspecies: Egretta rufescens dickey (van Rossem 1926): San Luis Island, Gulf of California.
Other names: Gaulin (Jamaica) in English; Garzón Rojiza, Garza Melenuda, Garza Piquirrosa, Garza Roja, Garza Rojiza, Garceta Rojiza in Spanish; Aigrette bleue, Aigrette roussâtre in French; Rötelreiher in German.
Description
The Reddish Egret is a medium dimorphic (dark or white) egret with a shaggy neck, and distinctive bicolored pink-and-black bill.
Adult: The dark morph adult has a red brown to dark cinnamon head and neck and slate grey to blue grey back, wings, tail, abdomen, and belly. White morph birds are entirely white. In both phases, the irises are straw yellow, set off by dark lores. The bill is dark toward the tip and dusky pink toward the head. Legs and feet are blue black to black.
During breeding, the head and neck become chestnut to pale cinnamon. The outer 45% of the bill becomes shiny black whereas the basal portion turns striking pink or flesh. The lores are turquoise blue during courtship changing to flesh during incubation and chick-rearing and then darkening toward the end of nesting. The shaggy lanceolate plumes of the head, neck and upper breast are cinnamon, while those of the back are grey mixed with some chestnut. In full development the filamentous back plumes extend up to 15 cm beyond the end of the tail, accentuating the bird’s characteristic shaggy appearance. The front of the legs and top of the feet remain black, but the sides and back of the legs and bottom of the feet become turquoise blue.
Variation: The sexes are alike, but males are slightly larger. The most important variation in the species is its white-dark dimorphism. The proportion of white birds differs in various populations, ranging from 5-12% in Texas, up to 15-20% in south Florida, 50% in Cuba, and almost 90% on Great Inagua in the Bahamas. Additionally, there is considerable individual variation in plumage shade of dark birds, which may be seasonal (R. Paul pers. comm.). Some dark birds have a number of white feathers, usually flight feathers on both sides or patches elsewhere (see photos in Hancock 2000). There are no reports of white birds with dark feathers. Geographic variation has been reported. Dickeyi from Baja California is slightly darker than rufescens (Payne 1979, Paul 1991).
Juvenile: The juvenile can be quite variable. Typically, the dark form is light grey brown above and paler on the belly. Feathers, such as wing coverts, are tipped cinnamon. Short head and breast plumes, first acquired at 4-6 months, are dull cinnamon. The bill is black to dark horn, usually lighter toward the base. The lores are black. Dark individuals range in base color from medium grey to grey brown often with a rufous tinge, some being quite rufous. Young white morph birds are all white. By 18 months juveniles resemble adults with worn plumage, and they attain full adult plumage in their third year.
Chick: The dark morph chick is covered with grey down with a cinnamon grey head crest. The white morph has white down.
VOICE: The Reddish Egret is usually silent. The “Raah” call is the flight and aggressive call. The “Arg” call is the advertising call. A soft “Awh” call, also rendered “crog-crog”, (R. Paul pers. comm.) is used the Greeting Ceremony and also in displays away from the nesting site. Bill Snaps are used in the Forward and Pursuit Flights. Chicks beg with raspy repeated call.
Weights and measurements: Length: 69-81 cm. Weight: 700-750 g.
Field characters
The Reddish Egret is identified by its shaggy appearance, two-toned bill during breeding, leg color, and its very active feeding behavior. The dark morph egret is distinguished from the adult Little Blue Heron by being larger and stouter with a thicker straight bill and shaggy lighter neck. The dark morph is distinguished from the Tricolor Heron by its dark (not white) belly, larger size and thicker appearance.
The white morph bird is distinguished from the immature Little Blue Heron by being larger, thicker, especially thicker neck, a heavier straight bill, dark (not green) legs, and its much more active feeding behavior. The white bird is distinguished from the Snowy Egret by being larger and stouter with a thicker bill, dark (not yellow) feet, two-toned bill (in breeding), and very active feeding behavior. It is distinguished from the Great Egret by being smaller, its two-tone (not yellow) bill, and active feeding behavior.
Systematics
The Reddish Egret is one of the medium egrets. Individual variation occurs among dark morph birds, which may be seasonal. Geographic variation has been recognized in two subspecies, with dickeyi being a weakly differentiated, slightly darker race (R. Paul pers. comm.).
Range and status
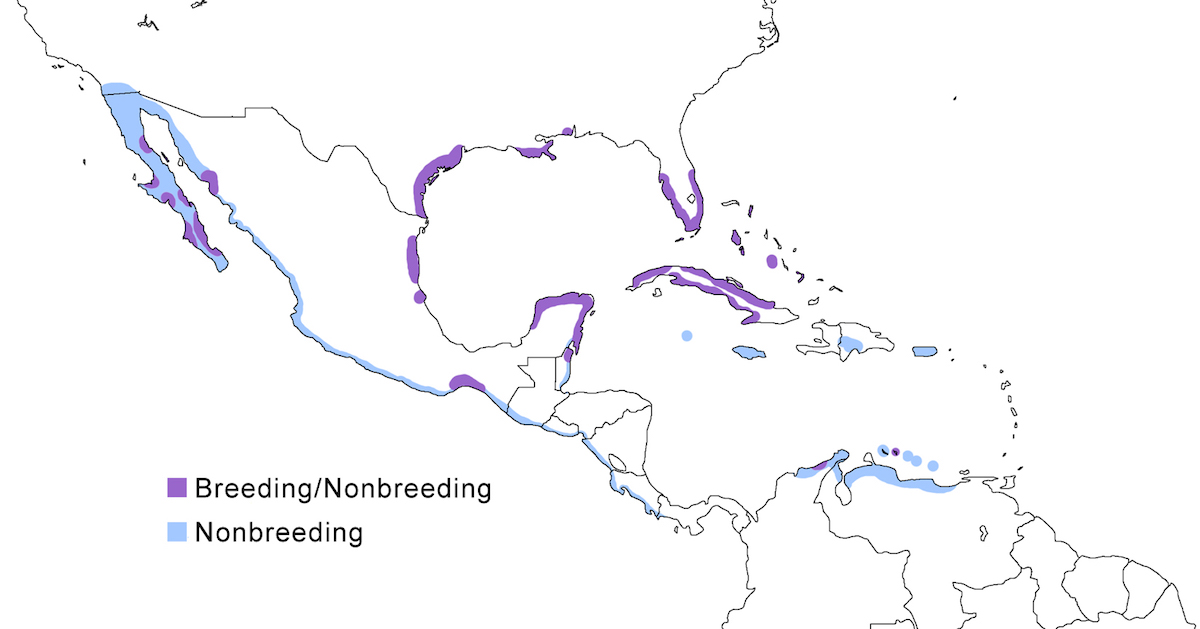
The Reddish Egret is a bird of the New World tropical coast from North America through the West Indies to northern South America.
Breeding range: This species breeds from east Florida (Merritt Island) and west Florida (Clearwater Harbor), coastal Gulf of Mexico (Alabama, Louisiana, Texas, east Mexico) to Belize (Lopez-Ornato and Ramo 1992), Baja California (Becerril and Carmona 1997), and west Mexico, Bahamas (Abaco, Grand Bahama, Andros, Acklins, Inagua), and Cuba (Garrido and Kirkconnell 1990, Denis et al. 1999). They also nest on Bonaire, Los Roques Venezuela, and likely on mainland Colombia and Isla Margarita (Scott and Carbonell 1986, Paul 1991). It is likely that the species breeds elsewhere than is presently known in Central America, the West Indies and north South America, but nesting records are nonexistent or not recent. Nesting is not documented for Central America, where few records exist (Paul 1991).
The race rufescens occurs over most of the range. The race dickeyi breeds in Baja California.
Nonbreeding range: Birds occur in winter in breeding areas along the Atlantic and Gulf Coasts from Florida, through Texas, and Mexico. They are increasingly found from north Florida to the Carolinas (R. Paul pers. comm.). On the Pacific Coast, birds winter north of the breeding area in south California (Roberson 1980), through west coastal Mexico. Scattered records occur in Central America. Birds occur year round on Cuba and the Bahamas, uncommonly on Cayman Island, Hispaniola and Puerto Rico. They occur with regularity on Venezuela’s offshore islands, Colombia, and Netherlands Antilles. There are scattered records for the Lesser Antilles (Norton 1993).
Migration: Florida and Texas populations are best considered to be partially migratory and also subject to some post-nesting dispersal and wandering. Distinguishing the two processes is not clear, but scattered Reddish Egret records, mostly of immature birds, have occurred in a number of localities in the United States in late summer, fall and even winter. Dispersal records include Massachusetts (Peterson 1992), New Jersey (Paxton et al. 1999), South Carolina (Wooten and Cox 1986), Kentucky, Indiana (Brock 1994), Kansas (Janzen 2000), Iowa (Schantz 1996), west Texas, Arizona (Benesh and Rosenberg 1998), Nevada (Kingery 1991). On the Pacific Coast Reddish Egrets have been reported in California and Arizona, USA. Individuals ringed in Texas have been found as far south as Campeche, Mexico, Guatemala, and El Salvador, likely crossing Mexico (Paul 1991).
Status: The species was adversely affected by plume hunting in the United States and extirpated from Florida in early 1900’s (Powell et al. 1989). It was recorded again in Florida in 1937 (Davis 1937, Desmond 1939) and since then has recolonized much of its former range (Paul et al. 1975, 1979, Rodgers and Schwikert 1986, Toland 1991). The Texas population decreased from 1930’s to 1970’s but recovered, with birds colonizing Louisiana and Alabama
Overall, both major North American populations increased and expanded their ranges in the past three of decades. However, more recently numbers in Texas may have declined, and there is serious concern for the future status of this population (Lange 1995, R. Paul pers. comm.). The documented breeding population in North America is about 2,600 pairs. The status of populations elsewhere is poorly known with only fragmentary information available about current nesting numbers. The Belize nesting population is about 100 pairs; the Inagua, Bahamas, population is 200 pairs. Overall, the documented population is about 3,000 pairs suggesting a total global population of no more than 5,000 pairs.
Habitat
The Reddish Egret is a bird of open marine flats and shorelines. Few are seen inland, and those are usually juveniles. Typically the species feeds on open, unvegetated tidal flats in very shallow water (less than 15 cm deep) (Paul 1991, 1996, Farmer 1992). It nests on coastal islands, usually low mangroves, bushes or cactuses.
Foraging
The Reddish Egret is one of the most active foragers among the herons. It feeds by Walking Slowly, Walking Quickly, Running, Hopping, Open Wing Feeding, Foot Raking, Underwing Feeding, Double Wing Feeding, and Hovering Stirring. It often uses Peering Over, probably in response to its highly reflective feeding habitat.
How it puts these behaviors together is characteristic. It runs across shallowly flooded flats, chasing schools of very small fish. It frequently changes directions, intermittently Hopping ahead, flapping wings as a disturbance, and finally stopping to stab when it catches up to prey. It feeds in very shallow water in open situations taking long strides, usually in a semi-crouch posture with head and neck extended forward. Reddish Egrets often run into the wind and sun. Open Wing Feeding is used most frequently during cloudy weather, in open water, and when the water surface was calm (Rodgers 1983).
They feed primarily alone, as would be required for such a wide-ranging foraging technique. Even in loose assemblages they tend to feed individually. When food becomes highly available, they do occur in large aggregations and multispecies flocks including other herons, pelicans (Pelecanus occidentalis), terns (Sterna), and stilts (Himantopus), spoonbills (Ajaia ajaja), and flamingos (Phoencopterus). In this situation they restrict their feeding behavior accordingly. It has been observed that the egrets sometimes gather into large highly compressed flocks, and it is possible that young of the year tend to assemble in flocks as an aid to foraging (Paul 1991), a hypothesis deserving testing.
Most prey items are marine and estuarine fish (McMurry 1971, Recher and Recher 1980, Paul 1991). Killifish dominate the diet, particularly minnows of the genus Cyprinodon, which has been identified as the main prey of the egret in Texas, Florida, and Bonaire. Other fish include mullet (Mugil), other killifish (Fundulus), topminnows (Poecilia) and pinfish (Lagodon). Shrimp and crabs are taken less frequently.
Breeding
Reddish Egrets nest in early spring, February–April on the Florida coasts, Florida Bay and on the Pacific coast. They nest later in spring, March to June, in Texas and the Gulf coast of Mexico, and in summer in Mexico. However, nesting timing seems to be variable and flexible across the range (Paul 1991). For example, nesting can be year-round in extreme south Florida, where there is also a fall nesting peak, November–January.
The Reddish Egret nests on protected islands near feeding sites, including man-made islands. Specific nesting substrate does not seem very important. They nest on mangroves (Rhizophora, Avicennia), bushes (Schinus, Baccharis), low herbaceous plants (Spartina, Sesuvium), cactus (Opuntia), or on the ground, oyster shell or sand. This is generally a colonial species, although they at times may nest solitarily. The nest is built of twigs lined with grasses and other fine vegetation.
The courtship behavior is complex and variable (Meyerriecks 1960, Palmer 1962). At the colony, the male claims a display site in the colony, which is first large and then reduced in size. It occupies and defends the site by Standing, Crest Raising, Upright (in which the body is uncharacteristically for this species sleeked, with crest raised only slightly), Forward, Pursuit Flights, and Face to Face Fighting from an upright posture. The Forward is quite aggressive; the feathers of head, neck and back fully erect, wings out, and the bird runs at the opponent giving the Raaah call. Pursuit Flights include Bill Snapping. Circle Flights and Pursuit Flights follow complex paths, the bird flying with its crest erected.
In the Stretch display, the bird extends its head fully upward, brings head slightly down, pits bill upward, erects head, back, breast plumes fully, bulges eyes, and performs Head Tossing moving head up and down. It also performs an Aerial Stretch in which it holds its head at a 45-degree angle while flying from a perch, Head Tossing and giving Crog call or Bill Snap. The Stretch and Head Tossing are used in a Greeting Ceremony and in twig passing.
There is much interaction among birds away from the nesting site, particularly near the onset of nesting. These involve birds interacting by Walking, Running or Hopping, with crest raised, Head Tossing, and giving the Crog call. An Aerial Stretch has been described on the feeding site, with the displaying bird flying around the other in a Circle Flight. Another feeding ground display is Peering Down, in which standing birds bend over with bills touching the water for up to a minute or two. Jumping Over is used by both birds on the feeding area. The jumping bird head tosses and erects its head, back and chest plumes. The functions of these interactions are not clear.
The most characteristic aspect of several displays is the inclusion of Head Tossing, rapidly raising and lowering its head with crest erected. This features the thick neck plumes and crest to maximum effect. Another common display is Crest Raising, in which the thick crest plumes are raised to make the head appear large.
The eggs are green blue, 47.6-52.9 x 34.6-38 mm. The usual clutch size is 2-4, but ranges to 7 (Paul 1991). Incubation is 26 days. Both parents attend young for about 3 weeks. Young leave the nest in 4-5 weeks. They remain nearby and return to the nest to feed. Young fly at 6-7 weeks, but are fed by the adults until 7-9 weeks (Paul 1991).
Hatching success is high overall, typically 69-85%, and survival from hatching to fledging is variable, averaging about 36% (Paul 1991). Lowered success may be caused by disturbance and food shortage. Reproduction was 1.2 to 1.5 young per pair in Texas and up to 1.8 young per pair in Florida, with lower figures occurring because of disturbance and food situations (Paul 1991).
Population dynamics
Some birds breed in their third year, but most in their fourth year (Paul 1991). This age at first breeding is unusually delayed for a medium-sized heron. Adult survival is not known.
Conservation
The Reddish Egret is globally vulnerable (Hafner et al. 2000). Its small global population size, small geographic range, and narrow foraging habitat requirements put this species at risk. Continuing urban and aquaculture development and alterations to coastal processes, upland runoff, or hydrography continue to threaten the suitability of these habitats for the species. Because it is a habitat specialist, the Reddish Egret has nowhere else to go when its habitats are lost. The near-impossible task of maintaining natural functioning in the coastal lagoons that it occupies is the primary conservation priority for the species. Sites of particular importance to these birds need to be identified and protected. Given that the maximum global population estimate is no more than 10,000 breeding birds, important areas are those supporting 100 or more birds at some time during the year. In the USA, the most important sites—such as Florida Bay, Tampa Bay, Laguna Madre, Galveston Bay, Aransas Bay, Corpus Christi Bay, Laguna Tamaulipas-need to be the focus of action plans that favour the species, including the management of hydrology and island nesting sites, and including the creation of new nesting sites where appropriate.
Research needs
Much remains to be known about the biology of this species, particularly clarifying its breeding behaviour and the function of display behaviour on the foraging grounds. Geographic variation in morphology, plumage, and biochemistry should be examined throughout the species’ range, especially thoroughly re-examining taxonomic status of the birds of Baja California and west Mexico. Surveys need to be conducted throughout the range to locate additional colony sites, and to catalogue locations qualifying as important areas for the species. Feeding habitat for major colonies should be identified and qualified as to potential threats. Nesting seasons throughout the range also need to be clarified. A cooperative multi-nation monitoring programme for important nesting areas should be developed. Production, survivorship, migration mortality, and other demographic parameters should be studied at several sites to assess the basic demography of the species. The concept of energy limitation and risk being a fundamental feature of this species’ ecology should be examined. Similarly, there should be additional study of the comparative foraging ecology of juvenile and adult egrets, including the role of dense feeding concentrations, to examine the role of habitat choice, flocking, and feeding effectiveness, particularly in light of this species’ delayed maturation. The wintering areas of the several populations of North American birds need to be better understood.
Overview
This is an intriguing bird. Its extravagant foraging technique must use a tremendous amount of energy, its prey are typically very small fish, and its efficiency of capture is low compared with other herons (Rodgers 1983). How its energy requirements are met working hard and inefficiently for small fish is a puzzle. Juveniles forage even more poorly. It is a species that has to learn how to feed well. Reddish Egrets can use their technique only in shallow, open habitats where deep water or vegetation will not impede their movements. But in such situations the predator is highly visible to the prey, thus the need to chase them down as they swim away from the disturbance the bird is causing. These are harsh environments, usually tidal and often periodically hypersaline, which is what restricts plant growth. The attraction of the Reddish Egret to such harsh environments is shown by its use of salt works (Paul 1991). In the places it chooses to forage, diversity of potential prey is not great, consisting of a few highly adaptable fish species. It eats only a few species of fish, and particularly concentrates on cyprinodontid killifish. Cyprinodon is found in shallow, coastal lagoons from fresh to hypersaline water throughout the egret’s range. The combination of habitat, foraging behaviour, and limited prey base is the essential element of the Reddish Egret’s feeding ecology. The species’ dependence on coastal tropical lagoon systems is the principal reason for its highly restricted range. It occurs in large numbers in few places. Given that, it seems likely that it occurs now in most of the places it is capable of using effectively.
This is a highly specialized heron. It has a limited habitat scope and depends on a single sort of prey. Given its specialization and energetically costly foraging style, nesting success and subadult survival must be tenuous. That it is able to recover from population crashes, shown twice, suggests that it has resilience. Likely adult mortality is low, especially for non-migratory birds. This is probably a long-lived species trading low reproductive potential within in its difficult environment for a longer adult lifespan. The possibility that resident and migratory birds coexist within a population suggests an even more complex demography. Adding to the complexity is the unknown function of dimorphism in this species. That the ratios of colours differ in different areas may be serendipity or evolutionarily meaningful. Its sexual maturity is delayed, twice as long as other medium herons, a feature likely related to the need to learn how to feed effectively and further suggesting the important role of adult survivorship in the species. The Reddish Egret is an intriguing species indeed.
§§§ Check out the Reddish Egret Working Group and its website to learn more and get involved with current conservation efforts.